Governance
This page contains information about the government structure for UKCRD, including a list of the delivery partners.
UK Clinical Research Delivery Programme’s (UKCRD) governance oversees, advises and implements the portfolio of work that the programme supports. While UKCRD has a set of key Delivery Partners that make up a majority of the governance groups, other organisations are often engaged with to ensure a true cross-sector approach.
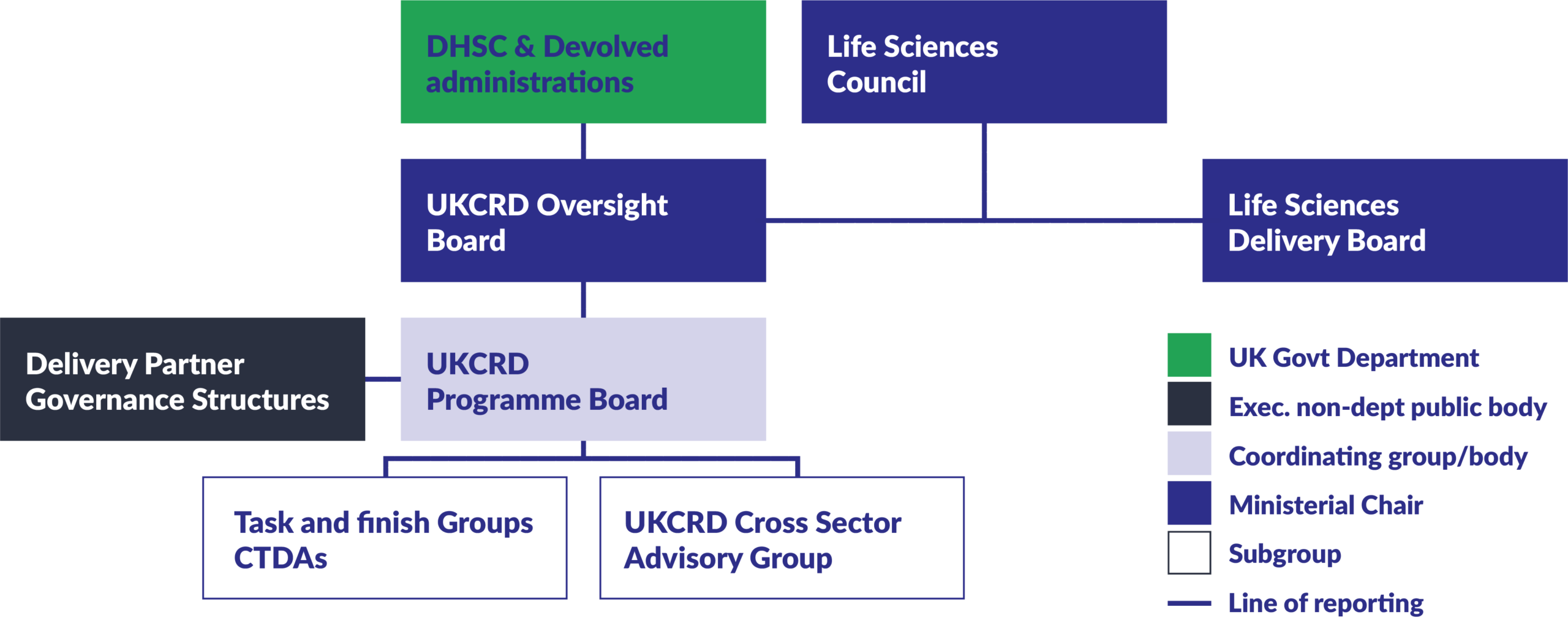
Governance groups
UKCRD Oversight Board
Oversees the UKCRD programme, taking decisions on strategic priorities and ensuring the coordination across programme partners. Co-chaired by Minister Ahmed and Lucy Chappell, Chief Scientific Adviser.
UKCRD Programme Board
Provides coordination and decision-making across organisations for the implementation of aims and priorities of the UKCRD Programme, to meet the aims of the programme, across the UK. Chaired by the Director of Science, Research and Evidence, DHSC. It brings together senior leaders of the programme’s partners to ensure collaborative delivery and alignment of strategic objectives.
UKCRD Programme Delivery partners include and are responsible for collaborating to deliver the workstreams that the UKCRD oversees, to achieve our aims of making the UK a world leader in clinical trials:
- Health and Care Research Wales (HCRW)
- Health and Social Care Northern Ireland (HSCNI)
- Health Research Authority (HRA)
- Medicines and Healthcare products Regulatory Agency (MHRA)
- National Institute for Health and Care Research (NIHR)
- NHS England (NHSE)
- NHS Research Scotland (NRS)
- Office for Life Sciences (OLS)
Cross Sector Advisory Group
Provides sound advice, support and guidance to the UKCRD Programme Board, to ensure the delivery and growth of a full portfolio of clinical research. Co-chaired by ABPI and AMRC.
The UKCRD programme places a strong emphasis on collaboration, bringing together a wide range of organisations from across the sector to ensure a comprehensive approach towards achieving the programmes ambitions for research across the UK. We actively welcome and value contributions from all stakeholders, whether commercial or non-commercial, ensuring that diverse perspectives and innovative ideas are heard and considered, to advise the UKCRD programme.
Cross Sector Advisory Group members include:
- Association of Medical Research Charities (AMRC) members charities
- Biopharmaceutical industry Association of British Pharmaceutical Industry (ABPI) members
- Health Research Authority (HRA)
- Medical Research Council (MRC)
- Medicines and Healthcare products Regulatory Agency (MHRA)
- NHS England (NHSE)
- NIHR Programmes
- NIHR Research Delivery Network (RDN), including separate Primary Care representation
- Patient and public representatives
- Representation from the Devolved Governments
- Royal College of Nursing (RCN)
- Royal College of Physicians (RCP)
- The research delivery workforce across NHS settings
- UK Research and Development (UKRD)
- UKCRC CTU Network
- University Hospitals Association
Task and Finish Groups
Groups that guide and advise on specific workstreams related to achieving the UKCRD’s aims and objectives. Chaired as required on a case-by-case basis. These are currently:
- Study Set-Up Plan Steering Group
- UK Clinical Research Inclusion Group
If you have any questions about the UKCRD governance structure, please get in touch via our contact page.
