Guidance on detailed definitions and processes for site-level performance reporting
Table of Contents
1. Introduction
Our UK performance for the set-up of commercial trials is important to our ability to compete for trials internationally and to offer new therapies to patients – through participation in trials and through marketed products based on trial evidence.
The Prime Minister has announced we must improve the speed of set-up for commercial clinical trials to 150 days or less, from regulatory submission to first patient, by March 2026.
The study-level metric only uses the data from the first site to recruit. But this requires all sites to focus on speed to make sure this ambition is met. In addition, the 150-day metric alone is not sufficient. All sites must focus on rapid set-up to increase the time available to recruit participants and recruit to time and target, as commercial sponsors expect, and to increase the number of participants in commercial clinical trials.
This guidance is focussed on the detailed definitions for time-points relating to set-up and recruitment for NHS/HSC sites. The guidance is focussed on clinical trials of investigational medicinal products for commercial contract research, which are the focus of the 150-day reporting. Further guidance on the 90-day metric, which has a broader scope, will expand the guidance to other types of research.
Sponsors and sites should use the definitions and processes set out in this document for all commercial contract CTIMPs, although certain studies will be excluded from national published reports (1).
This guidance is focussed on site metrics, and explains the data points for sites to collect, so that we can understand how to improve site set-up. Individual sites do not need to worry about the definitions of the components of the 150-day study-level metrics – these are calculated centrally from the site level reports. The definitions for each time point used are the same for both 150-day and 90-day metrics. The difference is in which timepoints are used to calculate the metrics and which site’s data is used.
Although it is likely that the lead site for a study should be the first site to set up and the first site with the first patient first visit, the 150-day metrics will use the first site achieving each of these timepoints, which may be at different sites. Therefore, most sites will not contribute to the 150-day metric. All sites should still aim to set up and recruit rapidly.
The guidance explains the correct sequence of activities for site set-up for both sponsors and sites, to ensure it is standardised and predictable for everyone. It gives new definitions of time points that have been developed based on feedback from sponsors and sites. Both sponsors and sites may have to update their SOPs and systems to comply. Sites need to make sure that they enter the right data matching these new definitions in their Local Portfolio Management System (LPMS).
The new definitions are designed to provide a fair reflection of site performance and take account of factors outside the site’s control. Some nations will specifically collect information on reasons outside the site’s control to understand national performance. Other sites may choose to collect information about causes of delay that helps them to understand their performance and to identify local improvement plans. Individual nations may use the information about reasons for delay to understand performance and may publish information about their national performance.
NIHR RDN will award enhanced funding to sites in England meeting performance metrics as set out in separate guidance. The reasons for delay will not be included in published reports or factored into any NIHR RDN performance-related funding in England.
Sites should look at guidance from their own nation on publication of reports and any additional funding awards. Performance data will use a range of the time points to understand performance so all data points recorded in their LPMS must match the definitions in this guidance.
Sites must submit all the data points requested in LPMS, otherwise it will not flow through to CPMS.
(1) Studies excluded from performance reporting are described in Appendix 3
2. Components of site-level metrics
Site-level metrics consist of two components:
Time for site set-up = 60 days or less
Time to recruit first participant = 30 days or less
All metrics are measured in calendar days.
3. Start of site set-up
Historically the start date for site set-up has been open to interpretation. This guidance clarifies the start date for the metric, and takes account of the different stages when sites are invited by sponsors.
Site invitation takes place prior to site selection. At site invitation, the sponsor provides the current version of the protocol or proposal, which may be an early draft. The sponsor and site should conduct a feasibility assessment to consider whether it has the relevant capability such as suitable staff expertise, equipment and facilities. If the sponsor agrees the site is suitable, the sponsor can choose to progress to select the site.
For all sites, site planning and set-up activities should start when the sponsor confirms the site is selected. No further feasibility assessment should be done after site selected.
The new commercial site selected letter of intent is a non-legally binding agreement between the commercial sponsor or Contract Research Organisation (CRO) and NHS organisation. It sets out agreement between the two parties that the NHS organisation has been selected as a site in the named study, based on the information described therein and, where relevant, attached to the email in which the Letter of Intent is sent. It is formal acknowledgement and agreement by both parties that the NHS organisation will have the capability, and that both parties will work to put in place the capacity, to deliver the study according to the timelines set out. The Letter of Intent should only be sent, and the NHS organisation formally selected as a site, following sponsor ‘due diligence’. This due diligence includes engaging appropriately with representatives of the Research and Development function and support departments of the site, in addition to the Principal Investigator (PI) and any relevant members of their research team, during site selection and feasibility. The Letter of Intent sets out the agreed pathway to site initiation, including any applicable steps prior to confirmation of capacity and capability or NHS management permission (Scotland).
The site selected letter of intent allows the sponsor to set either the date of receipt, or an alternative future date, as the date on which to start set-up activities.
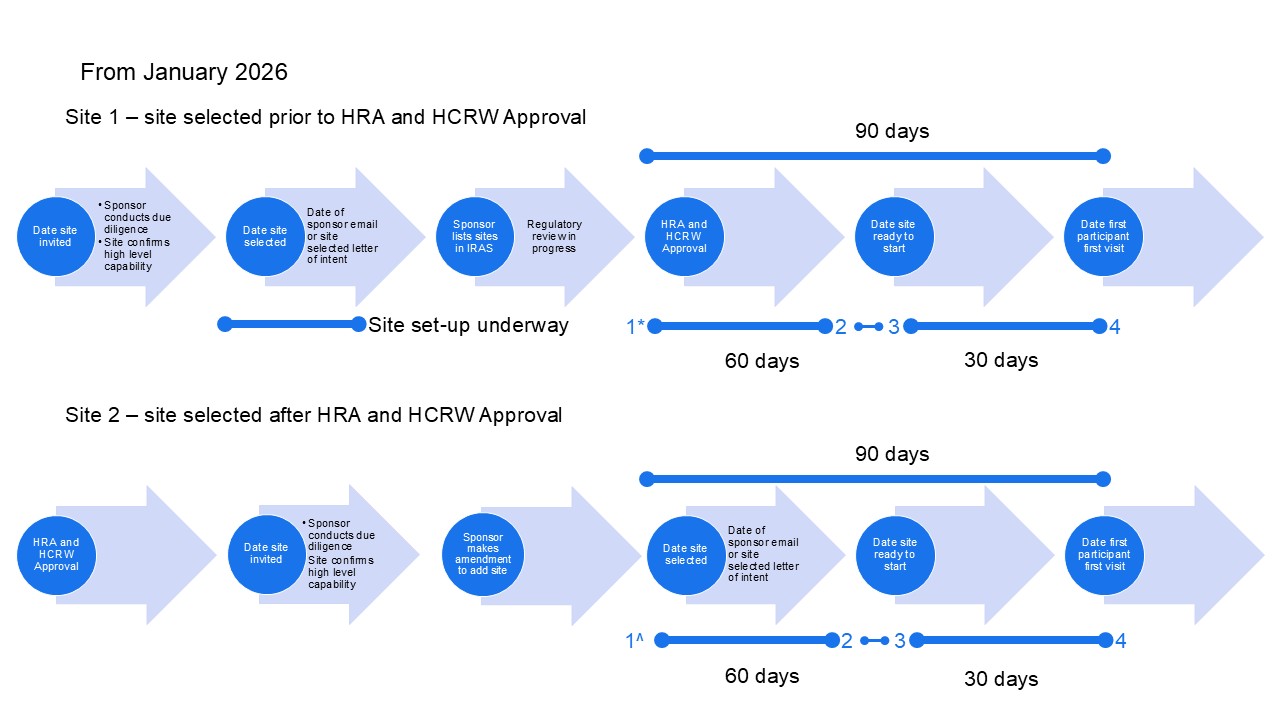
The start of site set-up activities is not necessarily the same as the metric used for reporting. The start of set-up activities is site selected, which is the date set in the commercial site selected letter of intent (or prior to full roll-out of the letter of intent, the email from the sponsor confirming site selected).
Sites selected prior to regulatory approval should start set-up activities in parallel to regulatory review using the draft documents available, when the site is selected.
Sites selected after regulatory approval should start site set-up activities immediately after having been selected, using the approved documents.
Sites selected during regulatory review of amendments should start site set-up activities immediately using the current documents (including any under review).
The Date Site Selected for reporting purposes has different definitions depending on the timing in relation to regulatory review.
For sites selected prior to regulatory approval, the Date Site Selected for reporting purposes is not the same as the start of set-up activities. In recognition that the time to regulatory approval is outside the site’s control, and that set-up activities are based on draft documents, the start of the reporting metric for these sites is the date of HRA and HCRW Approval in England and Wales, NRS Approval in Scotland and the equivalent completion of study-wide review in Northern Ireland. The start date is not the date of the combined review outcome, in recognition that sites are not able to confirm their capacity and capability until the completion of the study-wide review, including any other regulatory approvals. However, sites should not wait until regulatory approval before starting set-up and should start set-up from the site selected date, even though the metric used for reporting is the date of regulatory approval.
For sites selected after regulatory approval, the start of site set-up activities and the Date Site Selected for reporting purposes is the same. The start date for both set-up activity and reporting is the date set in the commercial site selected letter of intent (or prior to full roll-out of the letter of intent, the email from the sponsor confirming site selected).
For sites selected during regulatory review of amendments, the start of site set-up activities and the Date Site Selected for reporting purposes is the same. The start date for both set-up activity and reporting is the date set in the commercial site selected letter of intent (or prior to full roll-out of the letter of intent, the email from the sponsor confirming site selected).
4. End of site set-up
The Date Site Confirmed is the date of the last contract signature of all the organisations involved (sponsor, site, third party-if applicable) or date of NHS management permission in Scotland. Exchanging contracts and confirmation should not happen unless all steps required by the sponsor ahead of site initiation have been completed. That may include, for example, training, completion of logs, or pharmacy preparations, that the sponsor has stipulated at site selected. If there is any known delay in site initiation from the site (such as Principal Investigator availability) confirmation should not issued. Site confirmation always takes place after HRA and HCRW Approval (or NRS Approval or equivalent study-wide review in NI). As noted above, set-up activities should be undertaken in parallel to regulatory review.
The Date Site Ready to Start is the date the sponsor confirms green light to the site and the site is allowed to start recruiting.
Currently, the Date Site Confirmed is not used for reporting purposes.
End of the 60-day site set-up metric = Date Site Ready to Start
Start of the 30-day recruitment metric = Date Site Ready to Start
From January 2026, in recognition that the period between confirmation by the site and sponsor green light is outside the control of the site, the reporting metrics will change for the site metrics (not the study-level 150 days).
End of the 60-day site set-up metric = Date Site Confirmed
Start of the 30-day recruitment metric = Date Site Ready to Start
This new calculation will apply to all sites issuing confirmation of capacity and capability from January. For all sites confirming prior to January, and for NIHR RDN calculations for 2025, the end of the 60-day metric will be based on sponsor green light.
5. Time to recruitment
End of the 30-day recruitment metric = Date of First Participant First Visit
The Date of First Participant First Visit is the first date (in accordance with applicable study protocol and regulations) on which a potential participant signs an informed consent to participate in the trial. For interventional clinical trials this is the date of the first study visit of the first participant as set out in the protocol. In recognition that the site cannot control the eligibility of potential participants following post-consent screening, the date of the first study visit applies to the first participant for post-consent screening regardless of whether or not they are subsequently randomised.
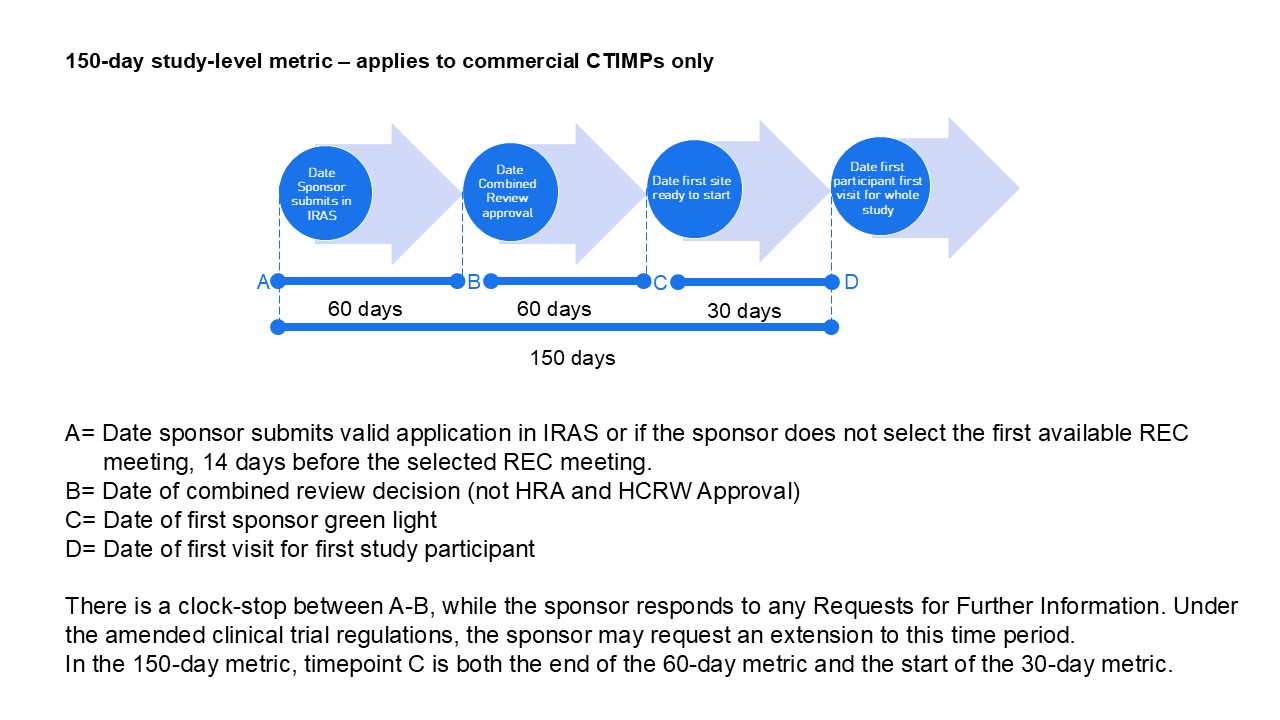
The 90-day metric is the sum of the separate 60-day and 30-day metrics. The sponsor may choose to delay initiating sponsor green light (stage 3 on the flowchart).
| 1* | HRA and HCRW Approval Date or equivalent |
| 1^ | Date Site Selected |
| 2 | Currently Date Site Ready to Start; from January 2026 Date Site Confirmed |
| 3 | Date Site Ready to Start |
| 4 | First Patient First Visit Date |
Definitions for timepoints
Numbers in the first column refer to flowcharts above. Each nation has published detailed guidance on matching these fields to LPMS fields. The first column shows the numbers matching the site-level diagrams.
| Current CPMS Data Field Label | New Definition | Further guidance | |
|---|---|---|---|
| 1* | HRA and HCRW Approval Date | The date the study receives HRA and HCRW Approval from England and Wales, NRS Approval from Scotland or equivalent completion of study wide review in Northern Ireland | |
| Date Site Invited | The date on the sponsor email received by the site providing the protocol in the version to be submitted for regulatory review, or a near final protocol. | This timepoint is not used in reports but must be completed for full data entry. | |
| 1^ | Date Site Selected | The date that the sponsor emails the site following an expression of interest and sponsor due diligence process, when the sponsor requests the site to start set-up. Where the sponsor chooses to identify potential sites early but set out a roll-out plan, the sponsor may choose to set a date when they expect the site to commence site set-up. | When the new site selected letter of intent is used, this metric will be the date that the sponsor sends this, or otherwise the data specified in the letter, on which set-up will commence, as agreed between the parties. Site set-up starts on receipt of this email, or otherwise on the date specified. |
| 2 | Date Site Confirmed | The date of the last contract signature of all the organisations involved (i.e. sponsor, site, third party-where applicable) or date of NHS management permission in Scotland. | Exchanging contracts and confirmation should not happen unless all steps required by the sponsor ahead of site initiation have been completed. |
| 3 | Date Site Ready to Start | The date the sponsor confirms green light to the site and the site is allowed to start recruiting. | |
| 4 | First Patient First Visit Date | The first date (in accordance with applicable study protocol and regulations) on which a potential participant signs an informed consent to participate in the trial. For interventional clinical trials this is the date (in accordance with the study protocol and regulations) of the first study visit of the first participant. | This will require a change in LPMS as currently a Consent Date for the First Recruited participant is sent to CPMS. This will need updating to First consent Date. If the participant is not randomised the first visit is still recorded. |
Appendices
Appendix 1
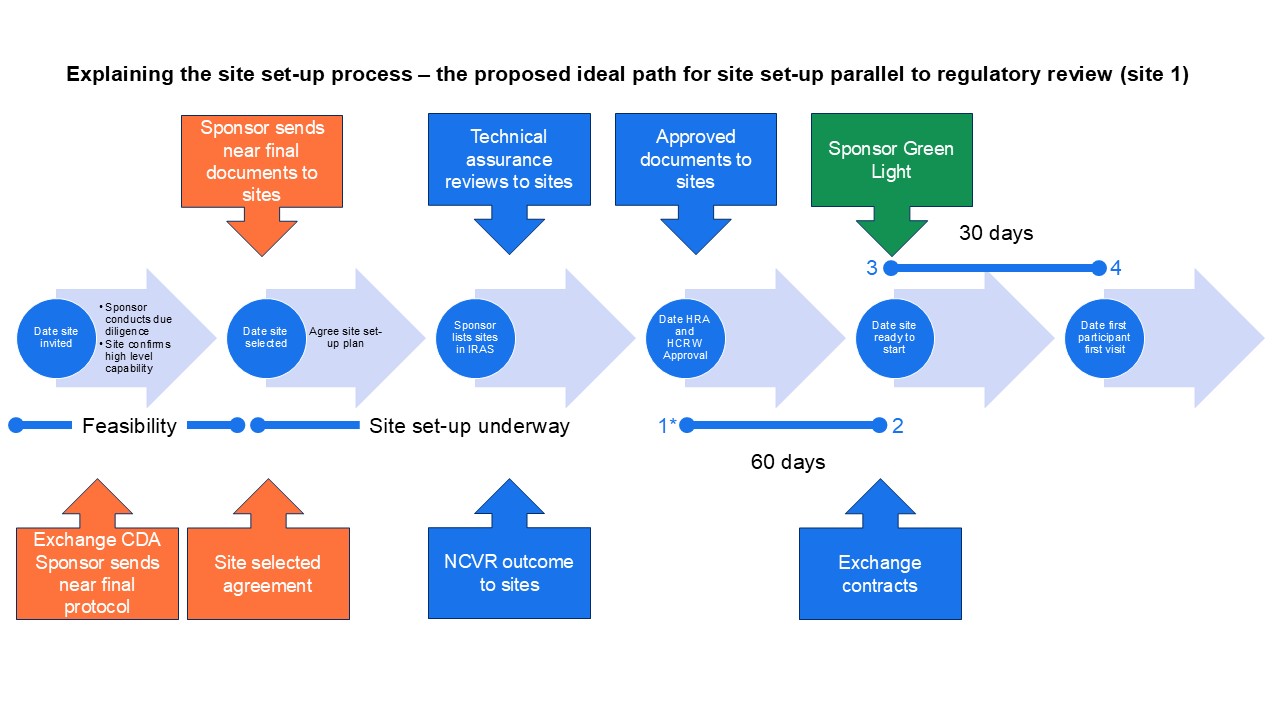
Detailed guidance for sites selected before regulatory approval (site 1 in diagrams)
Sites should complete set-up of commercial trials within 60 calendar days of HRA and HCRW Approval (or NRS Approval for Scotland or completion of study-wide review for NI). The site-level metric starts at HRA and HCRW Approval, not combined review outcome, because sites cannot issue confirmation until this date. Any delay between combined review and HRA and HCRW Approval is outside site control. Sites have 30 days from sponsor green light to recruit the first participant.
To best achieve the 60-day timeline:
Sites should start preparing from when they are selected as a site, in parallel to regulatory review, using the near final protocol and any available information provided by the sponsor. Site selected is the key decision. Once selected as a site, sites are no longer considering the feasibility of undertaking the study but are actively making arrangements to deliver it. This means that it is important to involve the right people from R&D, the delivery team and support departments in site selection visits and associated conversations. Technical assurance information may be sent to sites before the regulatory approved document pack is available, which will support set-up. Sites should accept the site price generated and not re-cost the study. Sites should conduct set-up in parallel to regulatory approvals and not wait for a full document pack, the local information pack or regulatory approval. Sites should aim to be ready to confirm readiness to deliver the study as soon after regulatory approval as possible.
From January 2026, the end of the 60-day timeline is the date site confirmed – the date of the last contract signature of all the organisations involved (i.e. sponsor, site, third party-as applicable) or date of NHS management permission in Scotland. Confirmation means everything is in place to start that the sponsor has requested, except for those things previously agreed by the sponsor to take place after contract exchange as part of the site initiation step.
To best achieve the 30-day timeline effectively:
The start date for the 30-day recruitment is the date the sponsor issues the green light to start. That means that, from January 2026, if there is a delay between site confirmed and sponsor green light, this time is not used in calculations because any delay is outside site control. Sites should pre-screen where appropriate and use the set-up phase to be as ready as possible to recruit participants after opening.
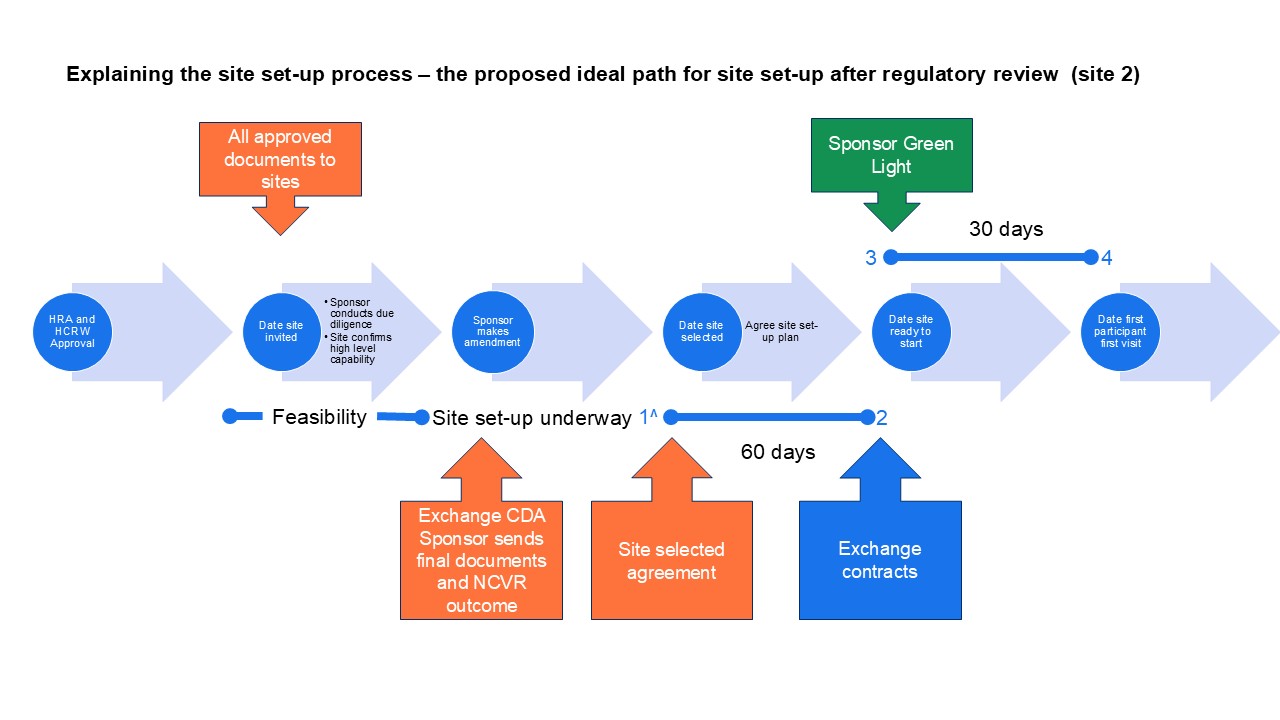
Detailed guidance for sites selected after regulatory approval (site 2 in diagrams)
If a site is selected for a commercial trial after the trial has received regulatory approval the site should aim to confirm it is ready by contract signature, within 60 days (nearly 9 weeks) of the sponsor confirming site selected by email. This is a new step that follows receipt of the regulatory documents and a discussion with the sponsor. Sites have 30 days from sponsor green light to recruit the first participant.
To best achieve the 60-day timeline:
Sites should start preparing from when they are selected as a site. Site selected is the key decision. Once selected as a site, sites are no longer considering the feasibility of undertaking the study but are actively making arrangements to deliver it. Unlike sites selected before regulatory approval these sites will have all the approved documents available to set up. This means that it is important to involve the right people from R&D, the delivery team and support departments in site selection visits and associated conversations. Sites should accept the site price generated and not re-cost the study. The end of the 60-day timeline is the date site confirmed – the date of the last contract signature of all the organisations involved (i.e. sponsor, site, third part-as applicable) or date of NHS management permission in Scotland. Confirmation means everything is in place to start, except for those things previously agreed by the sponsor to take place after contract exchange as part of the site initiation step. Exchanging contracts and confirmation should not happen unless all steps required by the sponsor ahead of site initiation have been completed.
To best achieve the 30-day timeline effectively:
The start date for the 30-day recruitment is the date the sponsor issues the green light to start. That means the clock stops if there is a delay between site confirmed and sponsor green light, because any delay is outside site control.
Sites should pre-screen where appropriate and use the set-up phase to be as ready as possible to recruit participants after opening.
Appendix 3
Dates should be entered for all timepoints for all study types. For published performance reports, some information will be excluded to avoid bias.
Exclusions from study-level 150-day metric
Study types excluded from reporting against the study-level 150-day metric by March 2026 are listed below:
- Observational, commercial collaborative and non-commercial studies.
- Studies where the 150-day metric is not expected to be met in the study milestone plan, such as rare disease and other low recruitment studies – using criteria that apply already in the UKCRD KPI Report – and extension studies. Definitions for these exclusions are listed here:
- ‘Rare disease’ definition: A rare disease is a life-threatening or chronically debilitating disease that affects 5 people or fewer in 10,000 of the UK population.
- ‘Low recruitment’ definition: Low recruitment refers to studies that – based on the UK sample size and proposed recruitment period – are expected to recruit fewer than 1 participant per month.
- ‘Extension study’ definition: Extension studies, or ‘roll-over studies,’ are studies that allow for patients participating in a parent clinical trial to ‘roll-over’ into a subsequent related study to continue to observe and measure long-term safety, tolerability, and/or effectiveness.
- Studies for which sponsors decide to opt out of the performance reporting. This is in recognition that sponsors may not wish to conduct their studies to this timeline, or may work to different processes.
Mechanisms to capture and inform sites about exclusions are in development. Further guidance will be provided around this.
Exclusions from site-level 90-day metric
For sites, there are elements that are outside the ability of a site to control and so they are excluded from published reports. For sites in England, these exclusions are also taken into account in payments from NIHR RDN as shown below. NIHR RDN performance-related funding applies to NHS Trusts.
| Studies excluded from site-level reporting | NIHR RDN financial implications in England | |
|---|---|---|
| 60-day metric | excludes extension studies | excluded from direct payment and performance adjustments |
| 30-day metric | excludes extension studies, rare disease studies and low-recruiting studies | excluded from performance adjustments |
| 90-day metric | excludes extension studies, rare disease studies and low-recruiting studies | excluded from direct payment |
Low-recruiting studies are defined in relation to the overall study sample size, not the individual site target, as this is not reliably available at national level.
The 90-day metric is calculated from the sum of the 60-day and 30-day metrics. Currently these time periods are continuous and so the 90-day metric is the same as the elapsed time from the start date to first patient first visit. From January 2026, the sum of the 60-day metric and the 30-day metric will not be the same as the total elapsed time due to the exclusion of the time between site confirmation and sponsor green-light.